COPPER FOIL
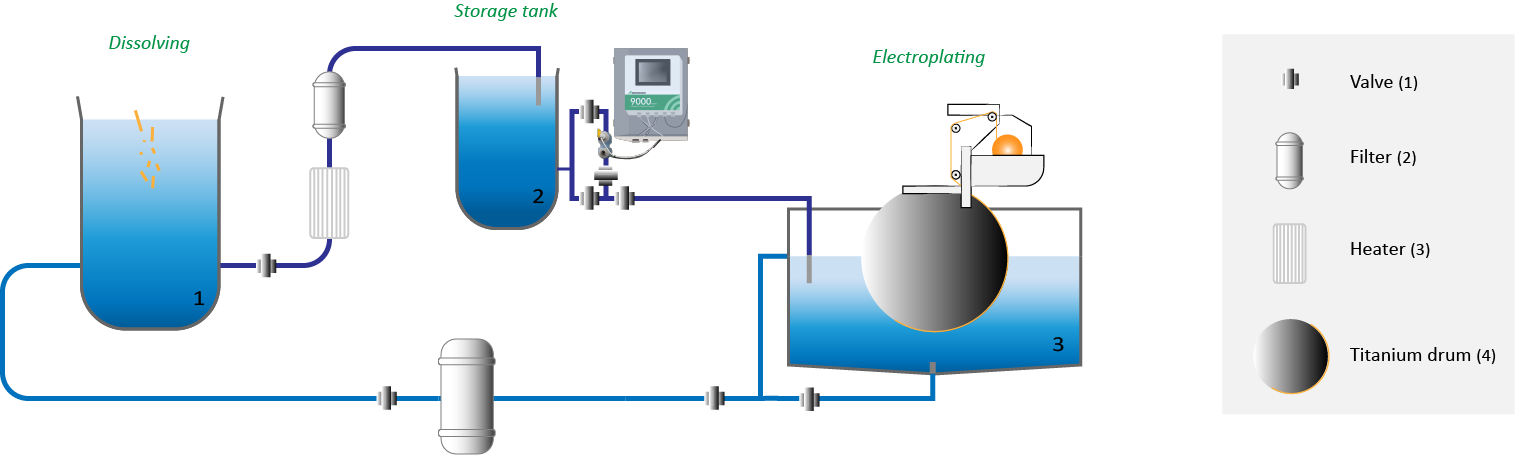
In the copper foil production process, one of the first stages is to dissolve high-grade (recycled) copper in Sulfuric Acid (H2S04)(1). This solution will pump through filters to produce the desired copper electrolyte solution and remove impurities. The last stage is a storage tank. This is where they keep the solution before its fed to the actual plating process to create copper foil(2).
On the side of this storage tank, in a recirculated bypass, they constantly monitor both concentrations (Cu + H2SO4). This is a critical measurement in the production process of copper foil. When the solution meets all chemical and quality standards, the solution is pumped into large plating tanks. The tanks contain partially immersed titanium drums to create a thin film of very pure copper. These are also referred to as electroplating (ED) machines(3).
The solution of dissolved Copper and Sulfuric Acid (H2SO4 & Cu++) is exactly monitored by the Rhosonics model CCM 9580 analyzer before the electroplating process. This model uses two sensors (ultrasonic & conductivity) to measure both concentrations in real-time through the bypass of the storage tank. The analyzer uses a predefined polynomial to calculate both concentrations. Besides continuous information about the two concentrations, the Rhosonics CCM 9580 also provides a real-time temperature reading.
In this way, the Rhosonics CCM 9580 model helps the client to maintain the desired concentrations to meet the quality standards for efficient copper foil production.
product choice for thE Copper foil industry
Get in touch with Rhosonics for further information. If you want to learn more about our applications, then please also check our client cases and webinars. For more information about Rhosonics or our ultrasonic analyzers, please take a look at our LinkedIn, Instagram, or Twitter page.